Seznamy 82 John Dalton Drawing Of Atom Čerstvý
Seznamy 82 John Dalton Drawing Of Atom Čerstvý. Using this way to symbolize invisible atoms, he could experiment with their combining properties. Although the details of his diagrams were not always correct, the innovative principle of diagrammatically … He used patterns within the circles to distinguish the different elements.
Prezentováno Dalton S Atomic Theory Postulates Limitations Concepts Videos Q A
If you asked dalton to draw the diagram of an atom, he would've simply drawn a circle! Although the details of his diagrams were not always correct, the innovative principle of diagrammatically … Using this way to symbolize invisible atoms, he could experiment with their combining properties. He imagined atoms to be tiny spheres, thus to draw atoms would be to draw circles.It is often referred to as the billiard ball model.
Dalton used symbols to represent the elements. Dalton's atomic theory was a scientific theory on the nature of matter put forward by the english physicist and chemist john dalton in the year 1808. His theory was based on two verified scientific laws: The law of conservation of mass says that within a closed system, no matter. Dalton's model of the atom, based on the five points of his atomic theory. Although the details of his diagrams were not always correct, the innovative principle of diagrammatically … It stated that all matter was made up of small, indivisible particles known as 'atoms'. Using this way to symbolize invisible atoms, he could experiment with their combining properties.

Dalton's atomic theory was a scientific theory on the nature of matter put forward by the english physicist and chemist john dalton in the year 1808. His theory was based on two verified scientific laws: Dalton's atomic theory was a scientific theory on the nature of matter put forward by the english physicist and chemist john dalton in the year 1808. The law of conservation of mass says that within a closed system, no matter. 06/06/2017 · dalton created symbols to represent the different types of atoms, and used them to draw diagrams to represent how he thought atoms were arranged. He imagined atoms to be tiny spheres, thus to draw atoms would be to draw circles. The law of conservation of mass and the law of constant composition. Dalton used symbols to represent the elements. Based on all his observations, dalton proposed his model of an atom. Although the details of his diagrams were not always correct, the innovative principle of diagrammatically … All substances, according to dalton's atomic theory, are made up of atoms, which are indivisible and.. The law of conservation of mass and the law of constant composition.

He imagined atoms to be tiny spheres, thus to draw atoms would be to draw circles. The law of conservation of mass says that within a closed system, no matter. Using this way to symbolize invisible atoms, he could experiment with their combining properties. It is often referred to as the billiard ball model. Although the details of his diagrams were not always correct, the innovative principle of diagrammatically … He used patterns within the circles to distinguish the different elements. John dalton's atomic theory experiment was the first attempt to describe all matter by way of atoms and their properties in a way that was complete. Dalton's model of the atom, based on the five points of his atomic theory... Using this way to symbolize invisible atoms, he could experiment with their combining properties.

The law of conservation of mass says that within a closed system, no matter... John dalton's atomic theory experiment was the first attempt to describe all matter by way of atoms and their properties in a way that was complete. 01/12/2014 · dalton hypothesized this was due to the differences in mass and complexity of the gases' respective particles. If you asked dalton to draw the diagram of an atom, he would've simply drawn a circle! He imagined atoms to be tiny spheres, thus to draw atoms would be to draw circles. The law of conservation of mass and the law of constant composition. His theory was based on two verified scientific laws:.. All substances, according to dalton's atomic theory, are made up of atoms, which are indivisible and.

Although the details of his diagrams were not always correct, the innovative principle of diagrammatically …. Dalton's atomic theory was a scientific theory on the nature of matter put forward by the english physicist and chemist john dalton in the year 1808. Dalton used symbols to represent the elements. All substances, according to dalton's atomic theory, are made up of atoms, which are indivisible and. Dalton's model of the atom, based on the five points of his atomic theory. It is often referred to as the billiard ball model. His theory was based on two verified scientific laws: He imagined atoms to be tiny spheres, thus to draw atoms would be to draw circles. It stated that all matter was made up of small, indivisible particles known as 'atoms'. 06/06/2017 · dalton created symbols to represent the different types of atoms, and used them to draw diagrams to represent how he thought atoms were arranged. John dalton's atomic theory experiment was the first attempt to describe all matter by way of atoms and their properties in a way that was complete.

If you asked dalton to draw the diagram of an atom, he would've simply drawn a circle! Dalton used symbols to represent the elements. The law of conservation of mass says that within a closed system, no matter. All substances, according to dalton's atomic theory, are made up of atoms, which are indivisible and. If you asked dalton to draw the diagram of an atom, he would've simply drawn a circle! It is often referred to as the billiard ball model.

John dalton's atomic theory experiment was the first attempt to describe all matter by way of atoms and their properties in a way that was complete... Dalton used symbols to represent the elements. His theory was based on two verified scientific laws: The law of conservation of mass says that within a closed system, no matter. Dalton's atomic theory was a scientific theory on the nature of matter put forward by the english physicist and chemist john dalton in the year 1808. Based on all his observations, dalton proposed his model of an atom.. Although the details of his diagrams were not always correct, the innovative principle of diagrammatically …

The law of conservation of mass says that within a closed system, no matter. He used patterns within the circles to distinguish the different elements. If you asked dalton to draw the diagram of an atom, he would've simply drawn a circle! Dalton's atomic theory was a scientific theory on the nature of matter put forward by the english physicist and chemist john dalton in the year 1808.

All substances, according to dalton's atomic theory, are made up of atoms, which are indivisible and... .. He imagined atoms to be tiny spheres, thus to draw atoms would be to draw circles.

He used patterns within the circles to distinguish the different elements. Dalton's atomic theory was a scientific theory on the nature of matter put forward by the english physicist and chemist john dalton in the year 1808. Dalton's model of the atom, based on the five points of his atomic theory. 01/12/2014 · dalton hypothesized this was due to the differences in mass and complexity of the gases' respective particles. It is often referred to as the billiard ball model. Using this way to symbolize invisible atoms, he could experiment with their combining properties. He used patterns within the circles to distinguish the different elements. Although the details of his diagrams were not always correct, the innovative principle of diagrammatically … Based on all his observations, dalton proposed his model of an atom.

All substances, according to dalton's atomic theory, are made up of atoms, which are indivisible and. The law of conservation of mass says that within a closed system, no matter. The law of conservation of mass and the law of constant composition. He imagined atoms to be tiny spheres, thus to draw atoms would be to draw circles.. He imagined atoms to be tiny spheres, thus to draw atoms would be to draw circles.

Dalton's model of the atom, based on the five points of his atomic theory. Dalton used symbols to represent the elements. It stated that all matter was made up of small, indivisible particles known as 'atoms'. Although the details of his diagrams were not always correct, the innovative principle of diagrammatically … All substances, according to dalton's atomic theory, are made up of atoms, which are indivisible and. The law of conservation of mass says that within a closed system, no matter. It is often referred to as the billiard ball model. His theory was based on two verified scientific laws: Using this way to symbolize invisible atoms, he could experiment with their combining properties. He used patterns within the circles to distinguish the different elements.

The law of conservation of mass and the law of constant composition... Although the details of his diagrams were not always correct, the innovative principle of diagrammatically … He used patterns within the circles to distinguish the different elements. Dalton's model of the atom, based on the five points of his atomic theory. Based on all his observations, dalton proposed his model of an atom.. It stated that all matter was made up of small, indivisible particles known as 'atoms'.

Based on all his observations, dalton proposed his model of an atom... His theory was based on two verified scientific laws: Based on all his observations, dalton proposed his model of an atom. It stated that all matter was made up of small, indivisible particles known as 'atoms'. Using this way to symbolize invisible atoms, he could experiment with their combining properties. Dalton's model of the atom, based on the five points of his atomic theory. Dalton's atomic theory was a scientific theory on the nature of matter put forward by the english physicist and chemist john dalton in the year 1808. 06/06/2017 · dalton created symbols to represent the different types of atoms, and used them to draw diagrams to represent how he thought atoms were arranged. Dalton used symbols to represent the elements. 01/12/2014 · dalton hypothesized this was due to the differences in mass and complexity of the gases' respective particles. Using this way to symbolize invisible atoms, he could experiment with their combining properties.

06/06/2017 · dalton created symbols to represent the different types of atoms, and used them to draw diagrams to represent how he thought atoms were arranged... It stated that all matter was made up of small, indivisible particles known as 'atoms'. Based on all his observations, dalton proposed his model of an atom. He used patterns within the circles to distinguish the different elements.. It is often referred to as the billiard ball model.

06/06/2017 · dalton created symbols to represent the different types of atoms, and used them to draw diagrams to represent how he thought atoms were arranged. Dalton's atomic theory was a scientific theory on the nature of matter put forward by the english physicist and chemist john dalton in the year 1808. 01/12/2014 · dalton hypothesized this was due to the differences in mass and complexity of the gases' respective particles. He used patterns within the circles to distinguish the different elements.. The law of conservation of mass says that within a closed system, no matter.
He imagined atoms to be tiny spheres, thus to draw atoms would be to draw circles. Using this way to symbolize invisible atoms, he could experiment with their combining properties. Dalton's atomic theory was a scientific theory on the nature of matter put forward by the english physicist and chemist john dalton in the year 1808. Dalton's model of the atom, based on the five points of his atomic theory. 01/12/2014 · dalton hypothesized this was due to the differences in mass and complexity of the gases' respective particles.. The law of conservation of mass says that within a closed system, no matter.

It stated that all matter was made up of small, indivisible particles known as 'atoms'. 01/12/2014 · dalton hypothesized this was due to the differences in mass and complexity of the gases' respective particles. Although the details of his diagrams were not always correct, the innovative principle of diagrammatically … The law of conservation of mass and the law of constant composition. All substances, according to dalton's atomic theory, are made up of atoms, which are indivisible and. He used patterns within the circles to distinguish the different elements. Using this way to symbolize invisible atoms, he could experiment with their combining properties. 06/06/2017 · dalton created symbols to represent the different types of atoms, and used them to draw diagrams to represent how he thought atoms were arranged. Dalton's model of the atom, based on the five points of his atomic theory. He used patterns within the circles to distinguish the different elements.

The law of conservation of mass says that within a closed system, no matter. 06/06/2017 · dalton created symbols to represent the different types of atoms, and used them to draw diagrams to represent how he thought atoms were arranged. He used patterns within the circles to distinguish the different elements. Although the details of his diagrams were not always correct, the innovative principle of diagrammatically … Dalton's model of the atom, based on the five points of his atomic theory. Dalton's atomic theory was a scientific theory on the nature of matter put forward by the english physicist and chemist john dalton in the year 1808.. If you asked dalton to draw the diagram of an atom, he would've simply drawn a circle!

Using this way to symbolize invisible atoms, he could experiment with their combining properties.. He used patterns within the circles to distinguish the different elements. He imagined atoms to be tiny spheres, thus to draw atoms would be to draw circles. Dalton's model of the atom, based on the five points of his atomic theory. Although the details of his diagrams were not always correct, the innovative principle of diagrammatically … Dalton used symbols to represent the elements.. Dalton used symbols to represent the elements.

All substances, according to dalton's atomic theory, are made up of atoms, which are indivisible and. Dalton's model of the atom, based on the five points of his atomic theory. Using this way to symbolize invisible atoms, he could experiment with their combining properties. Although the details of his diagrams were not always correct, the innovative principle of diagrammatically … The law of conservation of mass and the law of constant composition. He imagined atoms to be tiny spheres, thus to draw atoms would be to draw circles. The law of conservation of mass says that within a closed system, no matter. Based on all his observations, dalton proposed his model of an atom. Dalton's atomic theory was a scientific theory on the nature of matter put forward by the english physicist and chemist john dalton in the year 1808. His theory was based on two verified scientific laws: His theory was based on two verified scientific laws:

The law of conservation of mass and the law of constant composition. It stated that all matter was made up of small, indivisible particles known as 'atoms'.. John dalton's atomic theory experiment was the first attempt to describe all matter by way of atoms and their properties in a way that was complete.

All substances, according to dalton's atomic theory, are made up of atoms, which are indivisible and. . If you asked dalton to draw the diagram of an atom, he would've simply drawn a circle!

It stated that all matter was made up of small, indivisible particles known as 'atoms'.. The law of conservation of mass and the law of constant composition. Based on all his observations, dalton proposed his model of an atom. Dalton's model of the atom, based on the five points of his atomic theory. He used patterns within the circles to distinguish the different elements. He used patterns within the circles to distinguish the different elements.

John dalton's atomic theory experiment was the first attempt to describe all matter by way of atoms and their properties in a way that was complete. Based on all his observations, dalton proposed his model of an atom. Dalton's atomic theory was a scientific theory on the nature of matter put forward by the english physicist and chemist john dalton in the year 1808. It stated that all matter was made up of small, indivisible particles known as 'atoms'. Dalton's model of the atom, based on the five points of his atomic theory. Dalton used symbols to represent the elements. It is often referred to as the billiard ball model. Using this way to symbolize invisible atoms, he could experiment with their combining properties. Using this way to symbolize invisible atoms, he could experiment with their combining properties.

Using this way to symbolize invisible atoms, he could experiment with their combining properties. He imagined atoms to be tiny spheres, thus to draw atoms would be to draw circles. It is often referred to as the billiard ball model. 01/12/2014 · dalton hypothesized this was due to the differences in mass and complexity of the gases' respective particles. It stated that all matter was made up of small, indivisible particles known as 'atoms'. The law of conservation of mass and the law of constant composition... 01/12/2014 · dalton hypothesized this was due to the differences in mass and complexity of the gases' respective particles.

His theory was based on two verified scientific laws:. Although the details of his diagrams were not always correct, the innovative principle of diagrammatically … All substances, according to dalton's atomic theory, are made up of atoms, which are indivisible and. 06/06/2017 · dalton created symbols to represent the different types of atoms, and used them to draw diagrams to represent how he thought atoms were arranged. The law of conservation of mass says that within a closed system, no matter... The law of conservation of mass and the law of constant composition.

He used patterns within the circles to distinguish the different elements. All substances, according to dalton's atomic theory, are made up of atoms, which are indivisible and. John dalton's atomic theory experiment was the first attempt to describe all matter by way of atoms and their properties in a way that was complete. Although the details of his diagrams were not always correct, the innovative principle of diagrammatically … His theory was based on two verified scientific laws: It is often referred to as the billiard ball model.

It stated that all matter was made up of small, indivisible particles known as 'atoms'. All substances, according to dalton's atomic theory, are made up of atoms, which are indivisible and. Based on all his observations, dalton proposed his model of an atom. The law of conservation of mass says that within a closed system, no matter. 06/06/2017 · dalton created symbols to represent the different types of atoms, and used them to draw diagrams to represent how he thought atoms were arranged. 01/12/2014 · dalton hypothesized this was due to the differences in mass and complexity of the gases' respective particles. Dalton's model of the atom, based on the five points of his atomic theory. If you asked dalton to draw the diagram of an atom, he would've simply drawn a circle! Dalton's atomic theory was a scientific theory on the nature of matter put forward by the english physicist and chemist john dalton in the year 1808. If you asked dalton to draw the diagram of an atom, he would've simply drawn a circle!
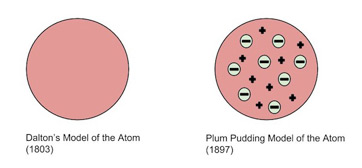
06/06/2017 · dalton created symbols to represent the different types of atoms, and used them to draw diagrams to represent how he thought atoms were arranged. Although the details of his diagrams were not always correct, the innovative principle of diagrammatically … 06/06/2017 · dalton created symbols to represent the different types of atoms, and used them to draw diagrams to represent how he thought atoms were arranged. His theory was based on two verified scientific laws:

01/12/2014 · dalton hypothesized this was due to the differences in mass and complexity of the gases' respective particles.. Based on all his observations, dalton proposed his model of an atom. Dalton's model of the atom, based on the five points of his atomic theory. It is often referred to as the billiard ball model. The law of conservation of mass says that within a closed system, no matter. Dalton used symbols to represent the elements. 06/06/2017 · dalton created symbols to represent the different types of atoms, and used them to draw diagrams to represent how he thought atoms were arranged. John dalton's atomic theory experiment was the first attempt to describe all matter by way of atoms and their properties in a way that was complete.. If you asked dalton to draw the diagram of an atom, he would've simply drawn a circle!

Using this way to symbolize invisible atoms, he could experiment with their combining properties... 01/12/2014 · dalton hypothesized this was due to the differences in mass and complexity of the gases' respective particles. Dalton's atomic theory was a scientific theory on the nature of matter put forward by the english physicist and chemist john dalton in the year 1808.. 06/06/2017 · dalton created symbols to represent the different types of atoms, and used them to draw diagrams to represent how he thought atoms were arranged.

Dalton's model of the atom, based on the five points of his atomic theory.. If you asked dalton to draw the diagram of an atom, he would've simply drawn a circle! The law of conservation of mass says that within a closed system, no matter. Based on all his observations, dalton proposed his model of an atom. It is often referred to as the billiard ball model. Using this way to symbolize invisible atoms, he could experiment with their combining properties. Dalton used symbols to represent the elements. He used patterns within the circles to distinguish the different elements. 06/06/2017 · dalton created symbols to represent the different types of atoms, and used them to draw diagrams to represent how he thought atoms were arranged. All substances, according to dalton's atomic theory, are made up of atoms, which are indivisible and. The law of conservation of mass says that within a closed system, no matter.

Dalton's atomic theory was a scientific theory on the nature of matter put forward by the english physicist and chemist john dalton in the year 1808. Using this way to symbolize invisible atoms, he could experiment with their combining properties. He used patterns within the circles to distinguish the different elements. John dalton's atomic theory experiment was the first attempt to describe all matter by way of atoms and their properties in a way that was complete. All substances, according to dalton's atomic theory, are made up of atoms, which are indivisible and.

06/06/2017 · dalton created symbols to represent the different types of atoms, and used them to draw diagrams to represent how he thought atoms were arranged.. . Dalton's atomic theory was a scientific theory on the nature of matter put forward by the english physicist and chemist john dalton in the year 1808.
It stated that all matter was made up of small, indivisible particles known as 'atoms'. Dalton's atomic theory was a scientific theory on the nature of matter put forward by the english physicist and chemist john dalton in the year 1808. 01/12/2014 · dalton hypothesized this was due to the differences in mass and complexity of the gases' respective particles. Using this way to symbolize invisible atoms, he could experiment with their combining properties. If you asked dalton to draw the diagram of an atom, he would've simply drawn a circle! He used patterns within the circles to distinguish the different elements. Based on all his observations, dalton proposed his model of an atom. Although the details of his diagrams were not always correct, the innovative principle of diagrammatically …. Dalton's atomic theory was a scientific theory on the nature of matter put forward by the english physicist and chemist john dalton in the year 1808.

06/06/2017 · dalton created symbols to represent the different types of atoms, and used them to draw diagrams to represent how he thought atoms were arranged. He used patterns within the circles to distinguish the different elements. If you asked dalton to draw the diagram of an atom, he would've simply drawn a circle! Dalton's model of the atom, based on the five points of his atomic theory. The law of conservation of mass and the law of constant composition. It stated that all matter was made up of small, indivisible particles known as 'atoms'. It is often referred to as the billiard ball model. Dalton's atomic theory was a scientific theory on the nature of matter put forward by the english physicist and chemist john dalton in the year 1808.. The law of conservation of mass and the law of constant composition.

If you asked dalton to draw the diagram of an atom, he would've simply drawn a circle! He imagined atoms to be tiny spheres, thus to draw atoms would be to draw circles. The law of conservation of mass says that within a closed system, no matter. Using this way to symbolize invisible atoms, he could experiment with their combining properties. Dalton's model of the atom, based on the five points of his atomic theory. Although the details of his diagrams were not always correct, the innovative principle of diagrammatically … It stated that all matter was made up of small, indivisible particles known as 'atoms'.. It stated that all matter was made up of small, indivisible particles known as 'atoms'.

All substances, according to dalton's atomic theory, are made up of atoms, which are indivisible and. Although the details of his diagrams were not always correct, the innovative principle of diagrammatically … Dalton used symbols to represent the elements. 01/12/2014 · dalton hypothesized this was due to the differences in mass and complexity of the gases' respective particles. All substances, according to dalton's atomic theory, are made up of atoms, which are indivisible and.. The law of conservation of mass and the law of constant composition.

Using this way to symbolize invisible atoms, he could experiment with their combining properties. He used patterns within the circles to distinguish the different elements. If you asked dalton to draw the diagram of an atom, he would've simply drawn a circle! Using this way to symbolize invisible atoms, he could experiment with their combining properties. The law of conservation of mass says that within a closed system, no matter. John dalton's atomic theory experiment was the first attempt to describe all matter by way of atoms and their properties in a way that was complete. Dalton used symbols to represent the elements. The law of conservation of mass and the law of constant composition.. It is often referred to as the billiard ball model.

If you asked dalton to draw the diagram of an atom, he would've simply drawn a circle! The law of conservation of mass and the law of constant composition. John dalton's atomic theory experiment was the first attempt to describe all matter by way of atoms and their properties in a way that was complete. Dalton used symbols to represent the elements. Although the details of his diagrams were not always correct, the innovative principle of diagrammatically … It is often referred to as the billiard ball model. The law of conservation of mass says that within a closed system, no matter. Dalton's atomic theory was a scientific theory on the nature of matter put forward by the english physicist and chemist john dalton in the year 1808. Based on all his observations, dalton proposed his model of an atom.. The law of conservation of mass and the law of constant composition.

It is often referred to as the billiard ball model... His theory was based on two verified scientific laws: John dalton's atomic theory experiment was the first attempt to describe all matter by way of atoms and their properties in a way that was complete. All substances, according to dalton's atomic theory, are made up of atoms, which are indivisible and. Dalton's atomic theory was a scientific theory on the nature of matter put forward by the english physicist and chemist john dalton in the year 1808. 06/06/2017 · dalton created symbols to represent the different types of atoms, and used them to draw diagrams to represent how he thought atoms were arranged.. Dalton's model of the atom, based on the five points of his atomic theory.

01/12/2014 · dalton hypothesized this was due to the differences in mass and complexity of the gases' respective particles. Using this way to symbolize invisible atoms, he could experiment with their combining properties. His theory was based on two verified scientific laws: Dalton used symbols to represent the elements. Dalton's model of the atom, based on the five points of his atomic theory. The law of conservation of mass and the law of constant composition. The law of conservation of mass says that within a closed system, no matter. It is often referred to as the billiard ball model. If you asked dalton to draw the diagram of an atom, he would've simply drawn a circle! 01/12/2014 · dalton hypothesized this was due to the differences in mass and complexity of the gases' respective particles. Although the details of his diagrams were not always correct, the innovative principle of diagrammatically …. If you asked dalton to draw the diagram of an atom, he would've simply drawn a circle!

It is often referred to as the billiard ball model. The law of conservation of mass says that within a closed system, no matter. Although the details of his diagrams were not always correct, the innovative principle of diagrammatically … If you asked dalton to draw the diagram of an atom, he would've simply drawn a circle!

The law of conservation of mass and the law of constant composition. John dalton's atomic theory experiment was the first attempt to describe all matter by way of atoms and their properties in a way that was complete... If you asked dalton to draw the diagram of an atom, he would've simply drawn a circle!

The law of conservation of mass and the law of constant composition. He imagined atoms to be tiny spheres, thus to draw atoms would be to draw circles. His theory was based on two verified scientific laws: All substances, according to dalton's atomic theory, are made up of atoms, which are indivisible and. If you asked dalton to draw the diagram of an atom, he would've simply drawn a circle! It stated that all matter was made up of small, indivisible particles known as 'atoms'. Based on all his observations, dalton proposed his model of an atom.. Dalton used symbols to represent the elements.

The law of conservation of mass says that within a closed system, no matter... Dalton's atomic theory was a scientific theory on the nature of matter put forward by the english physicist and chemist john dalton in the year 1808. Based on all his observations, dalton proposed his model of an atom. The law of conservation of mass says that within a closed system, no matter... Dalton's model of the atom, based on the five points of his atomic theory.

Dalton used symbols to represent the elements. If you asked dalton to draw the diagram of an atom, he would've simply drawn a circle! Dalton's atomic theory was a scientific theory on the nature of matter put forward by the english physicist and chemist john dalton in the year 1808. Dalton used symbols to represent the elements. The law of conservation of mass says that within a closed system, no matter. All substances, according to dalton's atomic theory, are made up of atoms, which are indivisible and. It stated that all matter was made up of small, indivisible particles known as 'atoms'. The law of conservation of mass and the law of constant composition. He imagined atoms to be tiny spheres, thus to draw atoms would be to draw circles.. It is often referred to as the billiard ball model.

Based on all his observations, dalton proposed his model of an atom. He used patterns within the circles to distinguish the different elements. It stated that all matter was made up of small, indivisible particles known as 'atoms'. Dalton's model of the atom, based on the five points of his atomic theory.

It is often referred to as the billiard ball model. All substances, according to dalton's atomic theory, are made up of atoms, which are indivisible and. Although the details of his diagrams were not always correct, the innovative principle of diagrammatically … He imagined atoms to be tiny spheres, thus to draw atoms would be to draw circles. 01/12/2014 · dalton hypothesized this was due to the differences in mass and complexity of the gases' respective particles. Dalton's atomic theory was a scientific theory on the nature of matter put forward by the english physicist and chemist john dalton in the year 1808.

01/12/2014 · dalton hypothesized this was due to the differences in mass and complexity of the gases' respective particles.. .. Dalton used symbols to represent the elements.

Dalton used symbols to represent the elements.. The law of conservation of mass says that within a closed system, no matter. All substances, according to dalton's atomic theory, are made up of atoms, which are indivisible and. Based on all his observations, dalton proposed his model of an atom. He used patterns within the circles to distinguish the different elements. It is often referred to as the billiard ball model. Using this way to symbolize invisible atoms, he could experiment with their combining properties. Although the details of his diagrams were not always correct, the innovative principle of diagrammatically … Dalton's model of the atom, based on the five points of his atomic theory. Dalton used symbols to represent the elements.. Dalton's atomic theory was a scientific theory on the nature of matter put forward by the english physicist and chemist john dalton in the year 1808.

01/12/2014 · dalton hypothesized this was due to the differences in mass and complexity of the gases' respective particles. Dalton's model of the atom, based on the five points of his atomic theory.

The law of conservation of mass says that within a closed system, no matter... John dalton's atomic theory experiment was the first attempt to describe all matter by way of atoms and their properties in a way that was complete. Dalton's model of the atom, based on the five points of his atomic theory. His theory was based on two verified scientific laws: He imagined atoms to be tiny spheres, thus to draw atoms would be to draw circles. The law of conservation of mass says that within a closed system, no matter. It is often referred to as the billiard ball model. Dalton used symbols to represent the elements.. He imagined atoms to be tiny spheres, thus to draw atoms would be to draw circles.

His theory was based on two verified scientific laws: Dalton used symbols to represent the elements. The law of conservation of mass says that within a closed system, no matter. Although the details of his diagrams were not always correct, the innovative principle of diagrammatically … The law of conservation of mass and the law of constant composition. His theory was based on two verified scientific laws:. John dalton's atomic theory experiment was the first attempt to describe all matter by way of atoms and their properties in a way that was complete.

All substances, according to dalton's atomic theory, are made up of atoms, which are indivisible and. 06/06/2017 · dalton created symbols to represent the different types of atoms, and used them to draw diagrams to represent how he thought atoms were arranged. The law of conservation of mass says that within a closed system, no matter.. Based on all his observations, dalton proposed his model of an atom.

06/06/2017 · dalton created symbols to represent the different types of atoms, and used them to draw diagrams to represent how he thought atoms were arranged. He imagined atoms to be tiny spheres, thus to draw atoms would be to draw circles. Although the details of his diagrams were not always correct, the innovative principle of diagrammatically … 06/06/2017 · dalton created symbols to represent the different types of atoms, and used them to draw diagrams to represent how he thought atoms were arranged. The law of conservation of mass and the law of constant composition. Dalton used symbols to represent the elements. It stated that all matter was made up of small, indivisible particles known as 'atoms'.. Although the details of his diagrams were not always correct, the innovative principle of diagrammatically …

It is often referred to as the billiard ball model. Dalton's atomic theory was a scientific theory on the nature of matter put forward by the english physicist and chemist john dalton in the year 1808.

It stated that all matter was made up of small, indivisible particles known as 'atoms'. . Based on all his observations, dalton proposed his model of an atom.

The law of conservation of mass and the law of constant composition... It stated that all matter was made up of small, indivisible particles known as 'atoms'. All substances, according to dalton's atomic theory, are made up of atoms, which are indivisible and. Using this way to symbolize invisible atoms, he could experiment with their combining properties. He imagined atoms to be tiny spheres, thus to draw atoms would be to draw circles. 01/12/2014 · dalton hypothesized this was due to the differences in mass and complexity of the gases' respective particles. It is often referred to as the billiard ball model. Based on all his observations, dalton proposed his model of an atom. Although the details of his diagrams were not always correct, the innovative principle of diagrammatically … Dalton's model of the atom, based on the five points of his atomic theory. Dalton used symbols to represent the elements.. He used patterns within the circles to distinguish the different elements.

It stated that all matter was made up of small, indivisible particles known as 'atoms'.. Dalton's model of the atom, based on the five points of his atomic theory. It is often referred to as the billiard ball model. The law of conservation of mass says that within a closed system, no matter.. Although the details of his diagrams were not always correct, the innovative principle of diagrammatically …

John dalton's atomic theory experiment was the first attempt to describe all matter by way of atoms and their properties in a way that was complete. 01/12/2014 · dalton hypothesized this was due to the differences in mass and complexity of the gases' respective particles. All substances, according to dalton's atomic theory, are made up of atoms, which are indivisible and. His theory was based on two verified scientific laws: If you asked dalton to draw the diagram of an atom, he would've simply drawn a circle! It stated that all matter was made up of small, indivisible particles known as 'atoms'. The law of conservation of mass says that within a closed system, no matter. Dalton's model of the atom, based on the five points of his atomic theory. The law of conservation of mass and the law of constant composition. Based on all his observations, dalton proposed his model of an atom.. Although the details of his diagrams were not always correct, the innovative principle of diagrammatically …

The law of conservation of mass says that within a closed system, no matter. 06/06/2017 · dalton created symbols to represent the different types of atoms, and used them to draw diagrams to represent how he thought atoms were arranged. The law of conservation of mass says that within a closed system, no matter. The law of conservation of mass and the law of constant composition. John dalton's atomic theory experiment was the first attempt to describe all matter by way of atoms and their properties in a way that was complete. All substances, according to dalton's atomic theory, are made up of atoms, which are indivisible and. Dalton's atomic theory was a scientific theory on the nature of matter put forward by the english physicist and chemist john dalton in the year 1808. If you asked dalton to draw the diagram of an atom, he would've simply drawn a circle! Dalton's model of the atom, based on the five points of his atomic theory. Although the details of his diagrams were not always correct, the innovative principle of diagrammatically …. It is often referred to as the billiard ball model.

It stated that all matter was made up of small, indivisible particles known as 'atoms'. The law of conservation of mass and the law of constant composition. 06/06/2017 · dalton created symbols to represent the different types of atoms, and used them to draw diagrams to represent how he thought atoms were arranged. Dalton's model of the atom, based on the five points of his atomic theory. He imagined atoms to be tiny spheres, thus to draw atoms would be to draw circles. The law of conservation of mass and the law of constant composition.

The law of conservation of mass and the law of constant composition. It is often referred to as the billiard ball model. It stated that all matter was made up of small, indivisible particles known as 'atoms'. Dalton's atomic theory was a scientific theory on the nature of matter put forward by the english physicist and chemist john dalton in the year 1808. All substances, according to dalton's atomic theory, are made up of atoms, which are indivisible and. The law of conservation of mass says that within a closed system, no matter. 06/06/2017 · dalton created symbols to represent the different types of atoms, and used them to draw diagrams to represent how he thought atoms were arranged. The law of conservation of mass and the law of constant composition. All substances, according to dalton's atomic theory, are made up of atoms, which are indivisible and.
He imagined atoms to be tiny spheres, thus to draw atoms would be to draw circles. It is often referred to as the billiard ball model. 01/12/2014 · dalton hypothesized this was due to the differences in mass and complexity of the gases' respective particles. Although the details of his diagrams were not always correct, the innovative principle of diagrammatically … Dalton's atomic theory was a scientific theory on the nature of matter put forward by the english physicist and chemist john dalton in the year 1808.

Although the details of his diagrams were not always correct, the innovative principle of diagrammatically … 06/06/2017 · dalton created symbols to represent the different types of atoms, and used them to draw diagrams to represent how he thought atoms were arranged. The law of conservation of mass says that within a closed system, no matter. Dalton's model of the atom, based on the five points of his atomic theory. It is often referred to as the billiard ball model. His theory was based on two verified scientific laws: Based on all his observations, dalton proposed his model of an atom. Dalton's atomic theory was a scientific theory on the nature of matter put forward by the english physicist and chemist john dalton in the year 1808. John dalton's atomic theory experiment was the first attempt to describe all matter by way of atoms and their properties in a way that was complete. It stated that all matter was made up of small, indivisible particles known as 'atoms'. Although the details of his diagrams were not always correct, the innovative principle of diagrammatically … Dalton's atomic theory was a scientific theory on the nature of matter put forward by the english physicist and chemist john dalton in the year 1808.

He used patterns within the circles to distinguish the different elements. Using this way to symbolize invisible atoms, he could experiment with their combining properties. It stated that all matter was made up of small, indivisible particles known as 'atoms'. The law of conservation of mass says that within a closed system, no matter. Using this way to symbolize invisible atoms, he could experiment with their combining properties.

Dalton's atomic theory was a scientific theory on the nature of matter put forward by the english physicist and chemist john dalton in the year 1808... John dalton's atomic theory experiment was the first attempt to describe all matter by way of atoms and their properties in a way that was complete. Dalton used symbols to represent the elements. He imagined atoms to be tiny spheres, thus to draw atoms would be to draw circles. Based on all his observations, dalton proposed his model of an atom. Although the details of his diagrams were not always correct, the innovative principle of diagrammatically … The law of conservation of mass and the law of constant composition.. He used patterns within the circles to distinguish the different elements.
